


Good news! The clinical verification results of two artificial intelligence technologies were published in the international well-known clinical journals of cardiac intervention
February 09, 2021
An important link for artificial intelligence technology to enter clinical applications is to be recognized by front-line clinical doctors. Recently, two innovative research works of Prof. Shengxian, Tu Academician Chen Yazhu's team, were published online in the journal EuroInterventio and Circulation: Cardiovascular Interventions. The two papers validate the accuracy of intravascular optical coherence tomography (IOCT) and Intravascular Ultrasound-Based Fractional Flow Reserve, which is the first time in the world to complete clinical transformation of OCT virtual histology technology and IVUS blood flow reserve fraction calculation technology. Both studies were also presented at EuroPCR 2020, as part of the newest clinical trial. The first authors of the paper are doctoral students Miao Chu and Wei Yu, and the corresponding author is Professor Shengxian Tu.

1. OCT virtual histological technique and clinical validation
Coronary artery atherosclerosis rupture is the main cause of the acute coronary syndrome. Comprehensive imaging assessment of plaque morphology in coronary arteries is beneficial to the early detection of high-risk lesions and the prevention of adverse cardiac events. Intravascular OCT imaging is the highest resolution imaging mode of living coronary artery imaging at present. The morphology of atherosclerotic plaques can be clearly observed and identified using OCT. However, in current clinical practice, the assessment of OCT images is mainly based on the visual observation of clinicians, which is highly subjective.
This study innovatively combines coronary anatomy features in the convolutional neural network to overcome the inherent shortcoming of the limited penetration depth of OCT. At the same time, it is suitable for different clinical centers, different quality of OCT images by using multi-center large-sample clinical data training network. In addition, the convolutional neural network can not only segment common atherosclerotic plaques but also identify inflammatory markers representing lesion instability, which is significant for the prediction of clinical events.
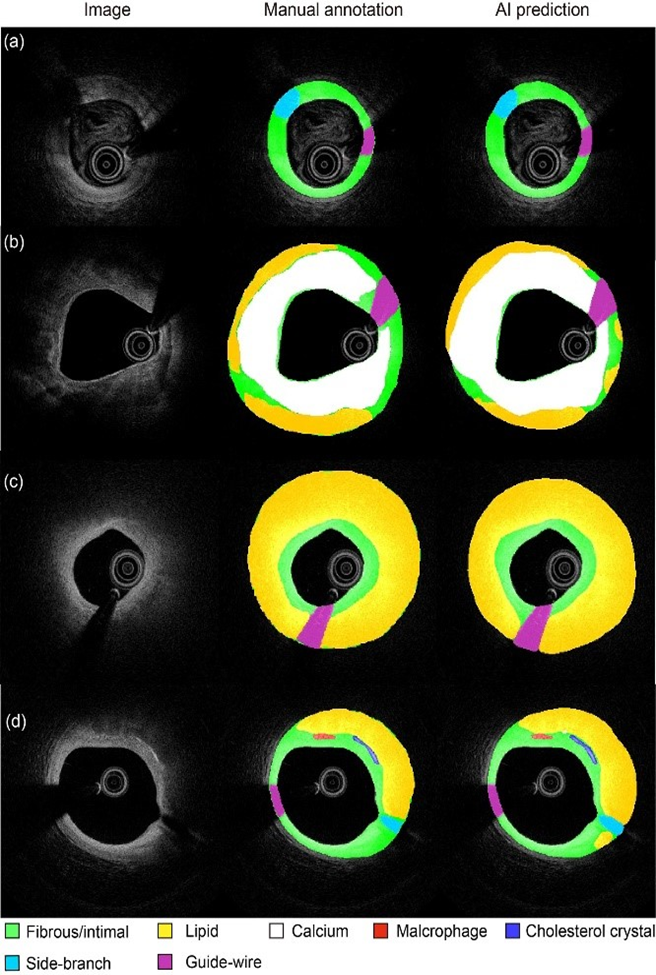
Figure 2 Segmentation in different challenging situations: images with suboptimal quality
A deep convolutional network for automatic plaque characterization with intravascular optical coherence tomography (IVOCT) was developed, using 509 pullbacks from 5 international centers. The model showed Dice coefficient 0.906 for fibrous plaque, 0.848 for calcium, and 0.772 for lipid, with excellent agreement in plaque burden quantification (R2=0.98). The model was externally validated on different datasets, using consensus among three international IVOCT core labs as reference. Diagnostic accuracy was 97.6% in fibrous plaque, 90.5% in lipid, and 88.5% in calcium. Artificial intelligence improves objectivity and reproducibility in the quantification of parameters of plaque composition, with potential applications in research and IVOCT guided PCI.
This work is supported by the National Key Research and Development Program of China, Natural Science Foundation of China [Grant Number 81871460, 82020108015, 81827806 and 81671763], and by Science Foundation Ireland Research Professorship Award [RSF 1413]. The DOI of this paper is 10.4244/EIJ-D-20-01355.
2. The measurement and Clinical verification of IVUS ultrasonic flow ratio(UFR)

Intravascular ultrasound imaging (IVUS) is a major method for intracavitary coronary artery imaging, which can clearly display the fine structure of coronary artery lesions. It has been widely used to optimize surgical strategies for PCI, especially for patients with complex lesions. At the same time, the effectiveness of the selection of revascularization regimens guided by FFR has also been widely confirmed in clinical practice. The combination of intracavitary imaging and FFR evaluation is a key standard of the current surgical planning of PCI. However, to realize the joint assessment of IVUS and FFR, two sets of assessment equipment and consumables need to be used at the same time, which will greatly increase the cost and operation process.
This paper proposed a novel method for fast computation of fractional flow reserve (FFR) from intravascular ultrasound images. The objective of this study is to evaluate the diagnostic performance of UFR using wire-based FFR as the reference. Post hoc computation of UFR was performed in consecutive patients with both intravascular ultrasound and FFR measurement in a core lab while the analysts were blinded to FFR. A total of 167 paired comparisons between UFR and FFR from 94 patients were obtained. Median FFR was 0.80 (interquartile range, 0.68–0.89) and 50.3% had a FFR≤0.80. Median UFR was 0.81 (interquartile range, 0.69–0.91), and UFR showed strong correlation with FFR (r=0.87; P<0.001). The area under the curve was higher for UFR than intravascular ultrasound-derived minimal lumen area (0.97 versus 0.89, P<0.001). The diagnostic accuracy, sensitivity, specificity, positive predictive value, negative predictive value, positive likelihood ratio, and negative likelihood ratio for UFR to identify FFR≤0.80 was 92% (95% CI, 87–96), 91% (95% CI, 82–96), 96% (95% CI, 90–99), 96% (95% CI, 89–99), 91% (95% CI, 93–96), 25.0 (95% CI, 8.2–76.2), and 0.10 (95% CI, 0.05–0.20), respectively. The agreement between UFR and FFR was independent of lesion locations (P=0.48), prior myocardial infarction (P=0.29), and imaging catheters (P=0.22). Intraobserver and interobserver variability of UFR analysis was 0.00±0.03 and 0.01±0.03, respectively. Median UFR analysis time was 102 (interquartile range, 87–122) seconds. Therefore, UFR had a strong correlation and good agreement with FFR. The fast computational time and excellent analysis reproducibility of UFR bears the potential of a wider adoption of integration of coronary imaging and physiology in the catheterization laboratory.
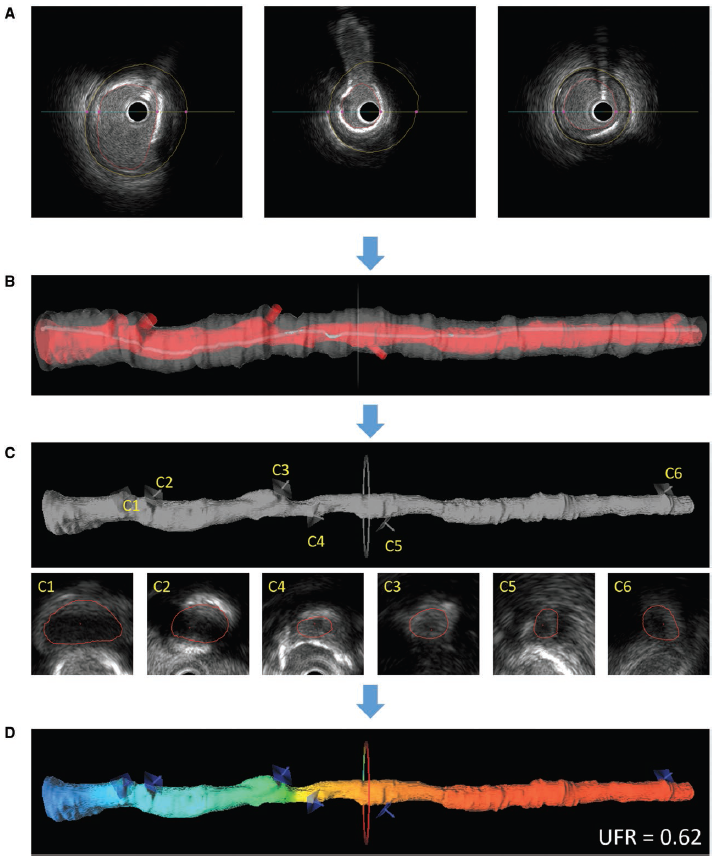
Figure 2: the calculation process of UFR
The cooperation units in this paper include Gifu Heart Center and National University of Ireland Galway in Ireland, etc. The research was supported by Shanghai Science and Technology Commission, etc.
Original paper link: https://www.ahajournals.org/doi/10.1161/CIRCINTERVENTIONS.120.009840